Boehler-Almax Sapphire Anvils
Alternative material to diamond
- Synthetic sapphire, Al2O3, Grade 1
- Substitute for diamonds in the pressure range up to about 20-25 GPa
- C-axis [001] orientation
- All surfaces are cut smooth and polished
Description
Product Number
| Diameter (mm)
| Material
| Application
| |
|---|---|---|---|---|
3.30 |
Sapphire |
Optical |
Material
Synthetic Sapphire is a single crystal form of corundum, Al2O3, also known as alpha-alumina, alumina, and single crystal Al2O3.
It may be used as a substitute for diamonds in the pressure range up to about 20-25 GPa.
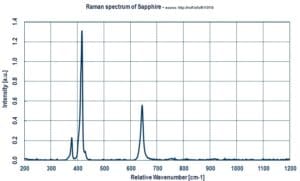
Sapphire has almost no peaks above 1000 cm−1 in the Raman spectrum and no absorption in the ultraviolet range. It is therefore especially useful for studies in those spectral regions.
Our synthetic sapphire is grade 1, which means it is free of insertions, block boundaries, twins, microbubbles and scattering centers.
Raman Spectrum
Application
The Gem Anvil Cell: high-pressure behaviour of diamond and related materials (Ji-An Xu, Ho-Kwang Mao and Russel J. Hemley, J. Phys.: Condens. Matter 14, 11549 (2002))
The moissanite anvil cell has been used to study the high-pressure behaviour of diamond. The first-order Raman shift of diamond shows a strong dependence on hydrostaticity, with very different pressure dependences observed under hydrostatic and non-hydrostatic conditions. The shift of the second-order Raman band under hydrostatic pressures was determined for the first time. Sapphire has almost no peaks above 1000 cm−1 in the Raman spectrum and no absorption in the ultraviolet range; it is therefore especially useful for studies in those spectral regions. A sapphire anvil cell was used in a study of graphite up to 24 GPa. A phase transition was found near 18 GPa, consistent with previous reports, and no peaks characteristic of diamond in the 1330 cm−1 range were found, indicating that the phase is not diamond.
FTIR Spectra of faceted diamonds and diamond simulants (Pimthing Thongnopkun, Sanong Ekgasit, Diamond and Related Materials 14, 1592 (2005))
FTIR spectra of faceted diamonds and diamond simulants collected by diffuse reflectance, transflectance, and specular reflection techniques were compared. The transflectance technique exploited total internal reflection phenomenon within the faceted diamond for the spectral acquisition. The transflectance spectra were similar to the well-accepted diffuse reflectance spectra with equal or better spectral qualities. Based on the observed spectral features of the faceted diamond, classification of the diamond, determination of defects, impurities, and treatment process (i.e., irradiation and high pressure and high temperature) can be performed.
High-pressure high-temperature Raman spectroscopy of kerogen: relevance of subducted organic carbon (Laurel Childress and Steven D. Jacobsen, Am.Mineralogist 102, 391 (2017))
The amount of insoluble macromolecular organic matter in the Earth’s crust, commonly referred to as kerogen, far exceeds the mass of living organic matter. The fraction of kerogen in sediments subducted into the mantle remains poorly constrained and will vary depending on the physical-chemical properties of kerogen along different slab geotherms. We studied the pressure-temperature evolution of carbon vibrational frequencies in isolated kerogen, previously not subjected to metamorphism, using Raman spectroscopy in a sapphire optical cell up to 3.2 GPa and 450 °C, corresponding to colder subduction geotherms. For blue-green laser excitation, we find optical irradiance exceeding ~3 kW/cm2 induces changes in spectral features of the primary graphitic (G-band) and two main disordered modes (D1 and D2) that might otherwise be mistaken for thermal maturation. Whereas previous in situ studies have investigated the changes in these molecular vibrations of kerogen at high temperature or high pressure, we collected Raman spectra of isolated kerogen at simultaneous high P-T conditions. Although instantaneous and irreversible changes in band ratios of isolated kerogen were observed above ~350 °C at room-pressure, long-duration (2–8 h) heating experiments at 450 °C and 2.7–3.0 GPa reveal no permanent change in band structure. The reduction in vibrational frequencies of the disordered carbon modes with temperature (dν/dT) at pressures >1 GPa is slightly less than found at room pressure, further indicating that pressure effectively increases the thermal stability of kerogen. Our results suggest that kerogen reaching depths of 60 km where the temperature is below ~450 °C may subduct into the mantle, providing a potential source for the organic-rich component of carbon recently detected in certain lower-mantle diamonds.
Documents
We have compiled a series of technical documents (brochures, articles, technical drawings, …) which you might find useful to help you understand this product better.
Articles
2017 – Childress, L.B. and Jacobsen, S.J. -High-pressure high-temperature Raman spectroscopy of kerogen: Relevance to subducted organic carbon
FAQs
How to chose the culet size ?
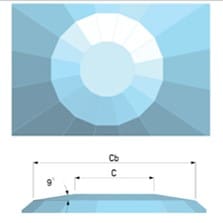
The culet size is to be decided as a function of the maximum pressure or the sample size. Typical sample space is given by the gasket thickness (typically 0.10 mm or less) and the central hole diameter (typically 30-50% culet size)
| Pmax (GPa) (*) | < 1 | 1-10 | 10-25 |
| Culet size (mm) | > 1.00 | 1-0.20 | < 0.20 mm, bevels up to 0.30 mm at 8° |
(*) The Pmax values are only indicative. The maximum pressure achievable with a DAC is influenced by many others experimental parameters, like the gasket characteristics (material, thickness and hole size) or the pressure transmitting medium.
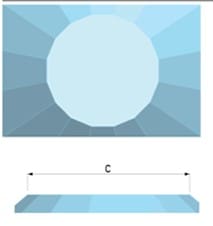
How the culet looks like?
| Culet flat | Culet with single bevel |
 |
 |
Examples of typical requests for quote:
Sapphire anvil, Diacell design, 16-sided, 2.50 mm, culet of 0.50 mm
- Article nr.: P01273
- Quantity: 6 anvils.
Sapphire anvil, Boehler Almax design, 8-sided, 3.30 mm – 85°, culet of 0.20 mm, bevels up to 0.30 at 8°
- Article nr.: P01274
- Quantity: 4 anvils.
You may also like…
Share this product on:










Reviews
There are no reviews yet.